Lithium ion conductivity, crystallization tendency, and microstructural evolution of LiZrxTi2-x(PO4)3 NASICON glass-ceramics (x = 0 - 0.4)
- 1 Ceramic Department, Materials and Energy Research Center (MERC), Alborz, Iran
- 2 School of Metallurgy & Materials Engineering, Iran University of Science and Technology, Tehran, Iran
Abstract
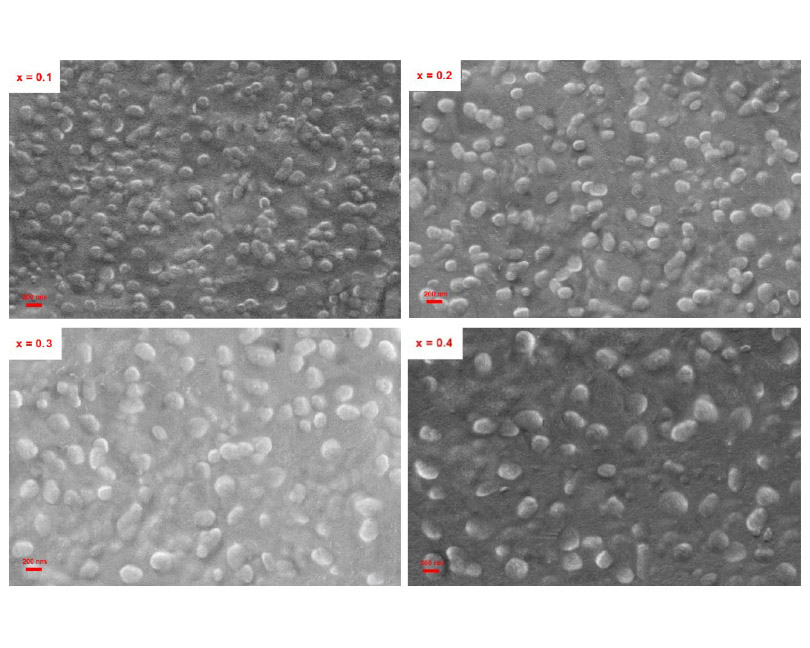
In this research, NASICON type (LiZrxTi2-x(PO4)3) glass-ceramics were fabricated (x = 0.1, 0.2, 0.3, and 0.4). Lithium-ion conductivity along with the crystallization tendency and microstructural features were examined in this regard. Parent glasses obtained through melt quenching were converted to the glass-ceramic specimens after one-step heat treatment procedure. The resultant glass-ceramics were deeply explored by means of different techniques including scanning electron microscope, differential thermal analysis, X-ray diffractometry, and ionic conductivity measurements. According to the obtained results, presence of Zr4+ ions in the glass network and its gradual increase caused the enhanced crystallization temperature as well as declined crystallinity and microstructure coarsening. In all studied glass-ceramics, LiT2(PO4)3 solid solution was the dominant crystalline phase and Zr4+ ions partly substituted in the structure of this crystalline phase. Moreover, presence of Zr4+ ions in the glass composition resulted in diminished lithium-ion conductivity of corresponded glass-ceramics at ambient temperature. Consequently, total conductivity of specimen with the highest level of ZrO2 (x = 0.4) was measured to be 0.78 x 10-5 Scm-1, being considerably less than ionic conductivity of the base (x = 0) glass-ceramic (3.04 x 10-5 Scm-1). It seems that less crystallinity of ZrO2 containing glass-ceramics decreases required connectivity between the lithium-ion free paths and is responsible for the diminished ionic conductivity of these specimens.
Downloads
References
Copyright (c) 2023 Parisa Goharian, Alireza Aghaei, Bijan Eftekhari Yekta, Sara Banijamali

This work is licensed under a Creative Commons Attribution 4.0 International License.
Copyright
Authors are the copyright holders of their published papers in Synthesis and Sintering, which are simultaneously licensed under a Creative Commons Attribution 4.0 International License. The full details of the license are available at https://creativecommons.org/licenses/by/4.0/.
All papers published open access will be immediately and permanently free for everyone to read, download, copy, distribute, print, search, link to the full-text of papers, crawl them for indexing, pass them as data to software, or use them for any other lawful purpose without any registration obstacles or subscription fees.